Abbott’s Navitor heart valve system just got a significant boost in Europe, with regulators expanding its approved use to treat patients at all surgical risk levels — a move that could dramatically increase treatment options for those suffering from severe aortic stenosis.
The medical device giant announced it received CE Mark approval for its Navitor transcatheter aortic valve implantation (TAVI) system to treat patients with symptomatic, severe aortic stenosis who are at low or intermediate risk for open-heart surgery. Previously, the device was only approved for high or extreme-risk patients in Europe, limiting its use to those most vulnerable.
A Breakthrough for Minimally Invasive Heart Valve Replacement
What makes this approval particularly noteworthy? The expanded indication is backed by impressive clinical results from the VANTAGE trial, which demonstrated a remarkably low 2.3% rate of all-cause mortality or disabling stroke at 12 months. Perhaps even more striking, the study showed no moderate or greater paravalvular leak — a common complication with valve replacements — at 30 days post-procedure.
“The VANTAGE study provides the scientific backbone for expanding Navitor’s indication to low- and intermediate-risk patients. The data are exceptional across both populations, confirming that the Navitor valve performs precisely as designed,” said Nicolas van Mieghem, MD, the trial’s principal investigator.
Aortic stenosis occurs when the heart’s aortic valve narrows, restricting blood flow and forcing the heart to work harder to pump blood throughout the body. Left untreated, this condition can lead to heart failure and death. Traditional treatment has often required open-heart surgery — a daunting prospect for many patients, especially older ones with other health concerns.
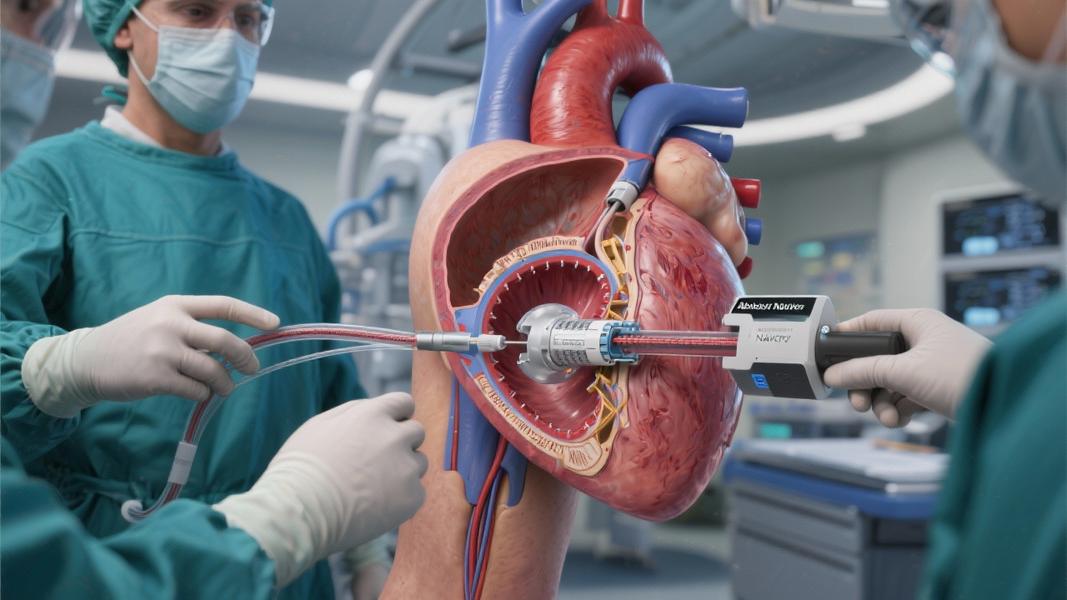
The Navitor system offers an alternative approach. Rather than cracking open the chest, doctors can deliver the replacement valve through a small incision in the leg, threading it up to the heart in a procedure that typically requires less recovery time and poses fewer risks for appropriate candidates.
Design Innovations That Address Long-standing Challenges
Abbott’s device isn’t just another entry in the increasingly crowded TAVI market. The Navitor features several design elements that set it apart from competitors, including a unique fabric cuff called NaviSeal that aims to reduce or eliminate the backflow of blood around the valve frame — a complication known as paravalvular leak that has plagued earlier generations of transcatheter valves.
Perhaps most forward-thinking is the valve’s positioning. “Up to 50% of younger patients with aortic stenosis will also get coronary artery disease in later years, and Navitor’s design preserves options and ability for lifetime disease management if future cardiac interventions are required,” Abbott notes in its announcement. This consideration could prove crucial as TAVI procedures move increasingly toward younger, lower-risk patients who may need additional heart treatments in the decades ahead.
Michael Reardon, MD, the Alison Family Distinguished Chair of Cardiovascular Research, has emphasized the device’s advancements: “Abbott’s Navitor device features advancements to help doctors safely and effectively treat patients with aortic stenosis, including a design that reduces the backflow of blood around the valve that’s often a complication following TAVI procedures.”
The system also utilizes Abbott’s FlexNav delivery system, which the company says ensures stable and accurate valve placement even in challenging anatomies — a critical factor for procedural success.
Part of a Broader Shift in Structural Heart Treatments
Is this expanded approval part of a larger trend? It certainly appears so. The announcement comes as European cardiology guidelines have also upgraded recommendations for other transcatheter therapies in Abbott’s portfolio. The European Society of Cardiology and European Association for Cardio-Thoracic Surgery have strengthened their endorsement of mitral and tricuspid valve transcatheter edge-to-edge repair therapies, including Abbott’s MitraClip and TriClip devices.
“Navitor is a strong example of how Abbott continues to evolve its structural heart portfolio to meet the growing demand for minimally invasive alternatives to open-heart surgery,” said Sandra Lesenfants, senior vice president of Abbott’s structural heart business. This evolution reflects both technological advancement and changing attitudes in cardiac care, where less invasive approaches are increasingly preferred when outcomes are comparable.
While the expanded CE Mark approval only affects European markets for now, it represents a significant milestone for Abbott’s TAVI program. In the United States, the Navitor system currently remains approved only for patients at high or extreme surgical risk, though this European expansion may foreshadow future regulatory developments stateside.
For the growing population of patients diagnosed with aortic stenosis — a condition that becomes more common with age — this expanded approval means more may avoid the trauma of open-heart surgery while still receiving effective treatment for a potentially deadly condition. That’s a heartening development, indeed.