European health regulators have approved a groundbreaking dissolving stent that could dramatically improve outcomes for millions suffering from a common but potentially devastating vascular disease.
Abbott announced today that its Esprit BTK Everolimus Eluting Resorbable Scaffold System has received CE Mark approval, making it available to the estimated 50 million Europeans living with peripheral artery disease (PAD) — a painful condition where arteries become clogged with plaque, preventing blood from reaching the lower extremities. For patients with the most severe form of PAD, the outlook has been grim, with just 25% surviving beyond ten years, according to Abbott officials.
A Disappearing Act That Could Save Limbs
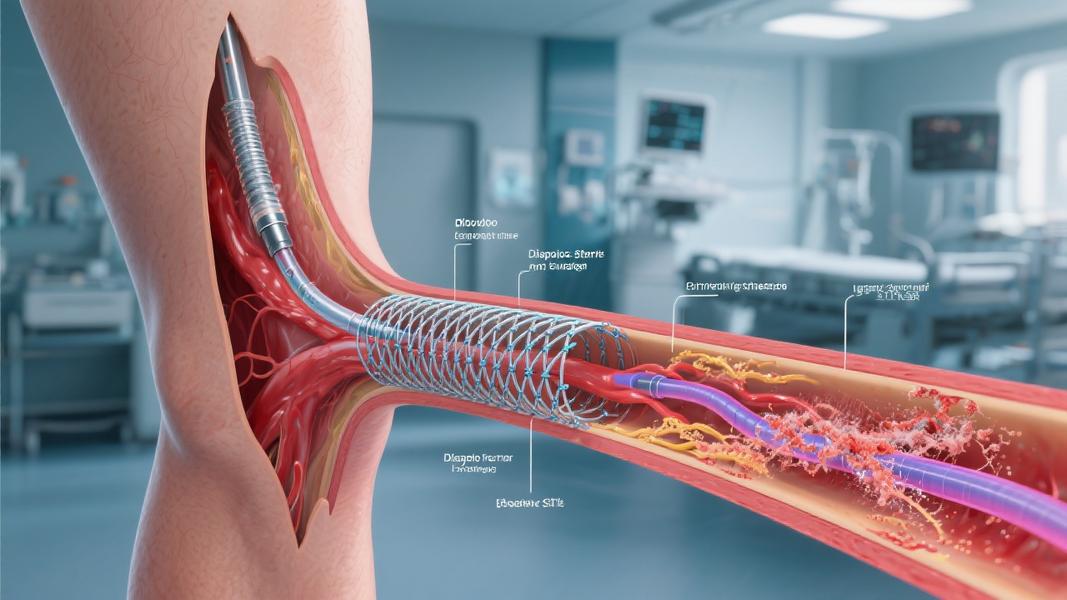
What makes this technology revolutionary? Unlike traditional metal stents that remain in the body permanently, the Esprit BTK System does something remarkable — it completely dissolves over time.
“The Esprit BTK System is designed to keep arteries open and deliver a drug called everolimus to support vessel healing prior to completely dissolving over time,” stated Abbott in its announcement. Made of material similar to dissolving sutures, the device is implanted through a minimally invasive procedure requiring only a small incision in the leg.
The scaffold provides temporary support to keep the artery open and prevent recoil while slowly releasing medication to promote healing. Then, over approximately three years, it gradually disappears completely from the body — a stark contrast to metal stents that remain permanently.
Clinical Evidence Shows Dramatic Improvement
Results from the LIFE-BTK clinical trial have shown the Esprit BTK System significantly outperforms the previous standard of care. Compared to balloon angioplasty, the dissolving scaffold reduced the need for repeat procedures by a substantial 48% and demonstrated 28.7-30.8% improvement in maintaining vessel patency and limb salvage at one and two years, data shows.
For patients with severe PAD below the knee, this represents a potential lifeline. “For too long, patients with severe PAD below the knee have had limited treatment options and were often faced with potential amputations,” said Professor Dierk Scheinert, M.D. “With CE Mark, the Esprit BTK System offers a resorbable scaffold backed by strong data and proven superiority over balloon angioplasty,” he explained.
The device already received FDA approval in April 2024, becoming the first drug-eluting resorbable scaffold for below-the-knee treatment in the United States. “The FDA approval of Abbott’s Esprit BTK System marks a significant milestone in our fight against peripheral artery disease below the knee and should usher in a new era of improved outcomes for people worldwide,” noted Sahil Parikh, director of endovascular services at Columbia University Irving Medical Center, in a statement.
Learning From Past Failures
But hasn’t this been tried before? Yes, and not successfully. Abbott’s previous venture into dissolving stent technology — the Absorb coronary stent — encountered unfavorable outcomes and was ultimately pulled from the market. What’s different this time?
Experts suggest the Esprit BTK device marks a turning point in bioresorbable scaffold technology. “Esprit BTK’s approval marks a turning point in bioabsorbable technology,” according to TCTMD. The device appears to be particularly suited for below-the-knee applications, where the clinical and biological benefits outweigh previous concerns.
The stakes couldn’t be higher. Peripheral artery disease at its most severe can lead to chronic limb-threatening ischemia, non-healing wounds, excruciating pain, and ultimately limb amputation. Many patients with advanced disease face limited options and poor quality of life.
“Abbott is an expert in the development of innovative treatments for vascular diseases, and pioneered dissolving stents for people with PAD below the knee,” said Samih Al Mawass, divisional vice president at Abbott EMEA. “Our resorbable program is focused on delivering meaningful innovation in the peripheral anatomy to help patients live healthier, fuller lives,” he added.
For the millions suffering from this debilitating condition across Europe, the approval represents something that’s been in short supply — hope that they might keep their limbs and their lives without permanent metal implants marking the battle.