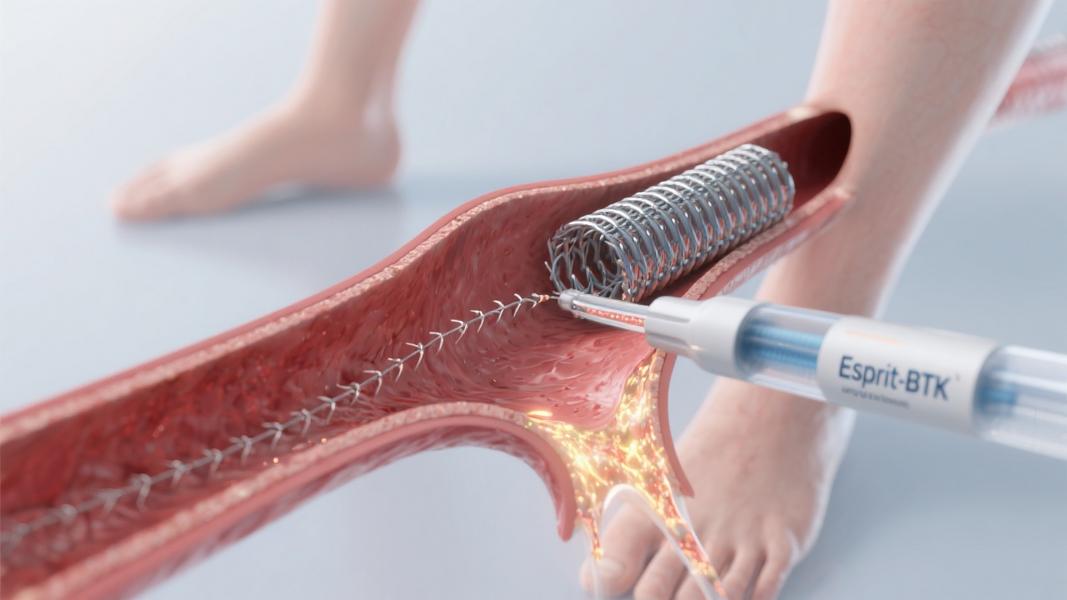
Abbott’s first-of-its-kind dissolving scaffold for blocked arteries below the knee has received CE Mark approval in Europe, offering new hope for millions suffering from severe peripheral artery disease (PAD). The Esprit BTK Everolimus Eluting Resorbable Scaffold System represents a significant breakthrough in vascular treatment technology that could reduce amputations and improve outcomes for patients with critical limb-threatening conditions.
The innovative device, which received U.S. FDA approval in April 2024 before gaining European clearance in 2025, functions similarly to a traditional stent but gradually dissolves after restoring blood flow. Made from a material akin to dissolving sutures, the scaffold “is designed to keep arteries open and deliver a drug called everolimus to support vessel healing prior to completely dissolving over time,” according to a statement from the company.
A Critical Need for Better Solutions
PAD affects approximately 50 million people in Europe — roughly twice the number in the United States. The condition causes arterial blockages that restrict blood flow to the lower limbs, often resulting in chronic pain, non-healing wounds, and in severe cases, amputation. The stakes couldn’t be higher: over a ten-year period, the most severe form of PAD has shown to have only a 25% survival rate, notes Abbott in its press materials.
What makes the Esprit BTK system so promising? Clinical data from the LIFE-BTK trial demonstrated that the device delivered “improved patient outcomes and 48% fewer repeat procedures over the study period” compared to standard balloon angioplasty, the company reports. These improvements in primary patency and limb salvage were sustained over two years of follow-up.
The scaffold is implanted via a minimally invasive catheter procedure and provides radial strength comparable to metallic stents within the first six months. It both supports vessel recoil and inhibits restenosis, then fully resorbs, “preserving future treatment options,” according to the company’s product information.
Learning from Past Setbacks
Abbott’s journey with bioresorbable technology hasn’t been without challenges. The company’s previous bioresorbable scaffold, Absorb, designed for coronary arteries, was withdrawn from markets worldwide by 2017 following safety concerns. But the Esprit BTK appears to have overcome those hurdles, demonstrating “improved clinical and biological efficacy without the prior issues of scaffold thrombosis,” as highlighted by industry observers.
Technically speaking, the new device consists of a bioresorbable poly-L lactide backbone with an everolimus coating and thin struts equivalent to contemporary coronary drug-eluting stents. In clinical trials, it achieved a procedural success rate of 91% versus just 70% with angioplasty alone, according to published data.
Professor Dierk Scheinert, a leading expert in vascular medicine, emphasized the device’s significance: “For too long, patients with severe PAD below the knee have had limited treatment options and were often faced with potential amputations… With CE Mark, the Esprit BTK System offers a resorbable scaffold backed by strong data and proven superiority over balloon angioplasty—giving physicians a novel, innovative tool to treat the most severe forms of PAD more effectively and improve patient outcomes across Europe,” he stated.
The Future of Vascular Treatment
Could this technology reshape treatment approaches for the millions suffering from severe PAD? Early indicators suggest it might. The ability to provide temporary structural support while delivering medication, then disappearing completely, represents a significant advancement over permanent metal implants.
Samih Al Mawass, divisional vice president of EMEA at Abbott’s vascular business, emphasized this point: “Abbott is an expert in the development of innovative treatments for vascular diseases, and pioneered dissolving stents for people with PAD below the knee… With the Esprit BTK System, we’re helping to restore blood flow without leaving a permanent implant behind,” he explained.
For patients with chronic limb-threatening ischemia — the most severe form of PAD — this approval couldn’t come soon enough. With limited effective options currently available and high rates of amputation and mortality, the Esprit BTK system represents not just a new device, but potentially a new lease on life for those suffering from this debilitating condition.